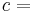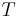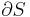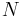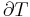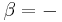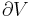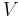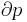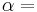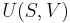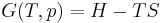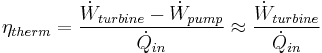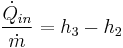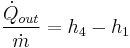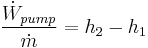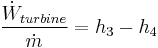Rankine cycle
The Rankine cycle is a cycle that converts heat into work. The heat is supplied externally to a closed loop, which usually uses water. This cycle generates about 90% of all electric power used throughout the world,[1] including virtually all solar thermal, biomass, coal and nuclear power plants. It is named after William John Macquorn Rankine, a Scottish polymath and Glasgow University professor. The Rankine cycle is the fundamental thermodynamic underpinning of the steam engine.
Contents |
Description
The Rankine cycle most closely describes the process by which steam-operated heat engines most commonly found in power generation plants generate power. The two most common heating processes used in these power plants are nuclear fission and the combustion of fossil fuels such as coal, natural gas, and oil.
The Rankine cycle is sometimes referred to as a practical Carnot cycle because, when an efficient turbine is used, the TS diagram begins to resemble the Carnot cycle. The main difference is that heat addition (in the boiler) and rejection (in the condenser) are isobaric in the Rankine cycle and isothermal in the theoretical Carnot cycle. A pump is used to pressurize the working fluid received from the condenser as a liquid instead of as a gas. All of the energy in pumping the working fluid through the complete cycle is lost, as is most of the energy of vaporization of the working fluid in the boiler. This energy is lost to the cycle because the condensation that can take place in the turbine is limited to about 10% in order to minimize blade erosion; the vaporization energy is rejected from the cycle through the condenser. But pumping the working fluid through the cycle as a liquid requires a very small fraction of the energy needed to transport it as compared to compressing the working fluid as a gas in a compressor (as in the Carnot cycle).
The efficiency of a Rankine cycle is usually limited by the working fluid. Without the pressure reaching super critical levels for the working fluid, the temperature range the cycle can operate over is quite small: turbine entry temperatures are typically 565°C (the creep limit of stainless steel) and condenser temperatures are around 30°C. This gives a theoretical Carnot efficiency of about 63% compared with an actual efficiency of 42% for a modern coal-fired power station. This low turbine entry temperature (compared with a gas turbine) is why the Rankine cycle is often used as a bottoming cycle in combined-cycle gas turbine power stations.
The working fluid in a Rankine cycle follows a closed loop and is reused constantly. The water vapor with entrained droplets often seen billowing from power stations is generated by the cooling systems (not from the closed-loop Rankine power cycle) and represents the waste energy heat (pumping and condensing) that could not be converted to useful work in the turbine. Note that cooling towers operate using the latent heat of vaporization of the cooling fluid. While many substances could be used in the Rankine cycle, water is usually the fluid of choice due to its favorable properties, such as nontoxic and nonreactive chemistry, abundance, and low cost, as well as its thermodynamic properties.
One of the principal advantages the Rankine cycle holds over others is that during the compression stage relatively little work is required to drive the pump, the working fluid being in its liquid phase at this point. By condensing the fluid, the work required by the pump consumes only 1% to 3% of the turbine power and contributes to a much higher efficiency for a real cycle. The benefit of this is lost somewhat due to the lower heat addition temperature. Gas turbines, for instance, have turbine entry temperatures approaching 1500°C. Nonetheless, the efficiencies of actual large steam cycles and large modern gas turbines are fairly well matched.
The four processes in the Rankine cycle
There are four processes in the Rankine cycle. These states are identified by numbers (in brown) in the diagram to the left.
- Process 1-2: The working fluid is pumped from low to high pressure. As the fluid is a liquid at this stage the pump requires little input energy.
- Process 2-3: The high pressure liquid enters a boiler where it is heated at constant pressure by an external heat source to become a dry saturated vapor. The input energy required can be easily calculated using mollier diagram or h-s chart or enthalpy-entropy chart also known as steam tables.
- Process 3-4: The dry saturated vapor expands through a turbine, generating power. This decreases the temperature and pressure of the vapor, and some condensation may occur. The output in this process can be easily calculated using the Enthalpy-entropy chart or the steam tables.
- Process 4-1: The wet vapor then enters a condenser where it is condensed at a constant temperature to become a saturated liquid.
In an ideal Rankine cycle the pump and turbine would be isentropic, i.e., the pump and turbine would generate no entropy and hence maximize the net work output. Processes 1-2 and 3-4 would be represented by vertical lines on the T-S diagram and more closely resemble that of the Carnot cycle. The Rankine cycle shown here prevents the vapor ending up in the superheat region after the expansion in the turbine, [1] which reduces the energy removed by the condensers.
Variables
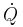 |
Heat flow rate to or from the system (energy per unit time) |
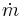 |
Mass flow rate (mass per unit time) |
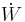 |
Mechanical power consumed by or provided to the system (energy per unit time) |
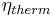 |
Thermodynamic efficiency of the process (net power output per heat input, dimensionless) |
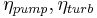 |
Isentropic efficiency of the compression (feed pump) and expansion (turbine) processes, dimensionless |
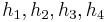 |
The "specific enthalpies" at indicated points on the T-S diagram |
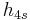 |
The final "specific enthalpy" of the fluid if the turbine were isentropic |
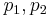 |
The pressures before and after the compression process |
Equations
In general, the efficiency of a simple Rankine cycle can be defined as:
Each of the next four equations[1] is easily derived from the energy and mass balance for a control volume. 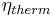 defines the thermodynamic efficiency of the cycle as the ratio of net power output to heat input. As the work required by the pump is often around 1% of the turbine work output, it can be simplified.
defines the thermodynamic efficiency of the cycle as the ratio of net power output to heat input. As the work required by the pump is often around 1% of the turbine work output, it can be simplified.
When dealing with the efficiencies of the turbines and pumps, an adjustment to the work terms must be made.
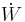 pump/
pump/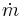 =
= 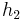 -
-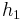 ≈
≈ 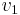 Δp/ηpump ≈
Δp/ηpump ≈ 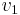 (
(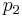 -
-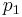 )/ηpump
)/ηpump
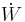 turbine/
turbine/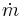 =
= 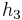 -
-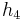 ≈ (
≈ (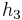 -
-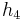 )*ηturbine
)*ηturbine
Real Rankine cycle (non-ideal)
In a real Rankine cycle, the compression by the pump and the expansion in the turbine are not isentropic. In other words, these processes are non-reversible and entropy is increased during the two processes. This somewhat increases the power required by the pump and decreases the power generated by the turbine.
In particular the efficiency of the steam turbine will be limited by water droplet formation. As the water condenses, water droplets hit the turbine blades at high speed causing pitting and erosion, gradually decreasing the life of turbine blades and efficiency of the turbine. The easiest way to overcome this problem is by superheating the steam. On the Ts diagram above, state 3 is above a two phase region of steam and water so after expansion the steam will be very wet. By superheating, state 3 will move to the right of the diagram and hence produce a drier steam after expansion.
Variations of the basic Rankine cycle
The overall thermodynamic efficiency (of almost any cycle) can be increased by raising the average heat input temperature 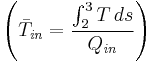 of that cycle. Increasing the temperature of the steam into the superheat region is a simple way of doing this. There are also variations of the basic Rankine cycle which are designed to raise the thermal efficiency of the cycle in this way; two of these are described below.
of that cycle. Increasing the temperature of the steam into the superheat region is a simple way of doing this. There are also variations of the basic Rankine cycle which are designed to raise the thermal efficiency of the cycle in this way; two of these are described below.
Rankine cycle with reheat
In this variation, two turbines work in series. The first accepts vapor from the boiler at high pressure. After the vapor has passed through the first turbine, it re-enters the boiler and is reheated before passing through a second, lower pressure turbine. Among other advantages, this prevents the vapor from condensing during its expansion which can seriously damage the turbine blades, and improves the efficiency of the cycle, as more of the heat flow into the cycle occurs at higher temperature.
Regenerative Rankine cycle
The regenerative Rankine cycle is so named because after emerging from the condenser (possibly as a subcooled liquid) the working fluid is heated by steam tapped from the hot portion of the cycle. On the diagram shown, the fluid at 2 is mixed with the fluid at 4 (both at the same pressure) to end up with the saturated liquid at 7. This is called "direct contact heating". The Regenerative Rankine cycle (with minor variants) is commonly used in real power stations.
Another variation is where bleed steam from between turbine stages is sent to feedwater heaters to preheat the water on its way from the condenser to the boiler. These heaters do not mix the input steam and condensate, function as an ordinary tubular heat exchanger, and are named "closed feedwater heaters".
The regenerative features here effectively raise the nominal cycle heat input temperature, by reducing the addition of heat from the boiler/fuel source at the relatively low feedwater temperatures that would exist without regenerative feedwater heating. This improves the efficiency of the cycle, as more of the heat flow into the cycle occurs at higher temperature.
Organic Rankine cycle
The organic Rankine cycle (ORC) uses an organic fluid such as n-pentane[2] or toluene[3] in place of water and steam. This allows use of lower-temperature heat sources, such as solar ponds, which typically operate at around 70–90 °C.[4] The efficiency of the cycle is much lower as a result of the lower temperature range, but this can be worthwhile because of the lower cost involved in gathering heat at this lower temperature. Alternatively, fluids can be used that have boiling points above water, and this may have thermodynamic benefits. See, for example, mercury vapour turbine.
The Rankine cycle does not restrict the working fluid in its definition, so the inclusion of an “organic” cycle is simply a marketing concept that should not be regarded as a separate thermodynamic cycle.
References
- ^ Wiser, Wendell H. (2000). Energy resources: occurrence, production, conversion, use. Birkhäuser. p. 190. ISBN 9780387987446. http://books.google.com/books?id=UmMx9ixu90kC&pg=PA190&dq=electrical+power+generators+steam+percent&hl=en&ei=JppoTpVexNmBB4C72MkM&sa=X&oi=book_result&ct=result&resnum=2&ved=0CDgQ6AEwATgK#v=onepage&q=steam&f=false.
- ^ Canada, Scott; G. Cohen, R. Cable, D. Brosseau, and H. Price (2004-10-25). "Parabolic Trough Organic Rankine Cycle Solar Power Plant". 2004 DOE Solar Energy Technologies (Denver, Colorado: US Department of Energy NREL). http://www.nrel.gov/csp/troughnet/pdfs/37077.pdf. Retrieved 2009-03-17.
- ^ Batton, Bill (2000-06-18). "Organic Rankine Cycle Engines for Solar Power". Solar 2000 conference. Barber-Nichols, Inc.. http://www.nrel.gov/csp/troughnet/pdfs/batton_orc.pdf. Retrieved 2009-03-18.
- ^ Nielsen et al., 2005, Proc. Int. Solar Energy Soc.
- ^Van Wyllen 'Fundamentals of thermodynamics' (ISBN 85-212-0327-6)
- Moran & Shapiro 'Fundamentals of Engineering Thermodynamics' (ISBN 0-471-27471-2)
- Wikibooks Engineering Thermodynamics
|
||||||||||||||||||||||
|
|||||||||||||||||||||||||